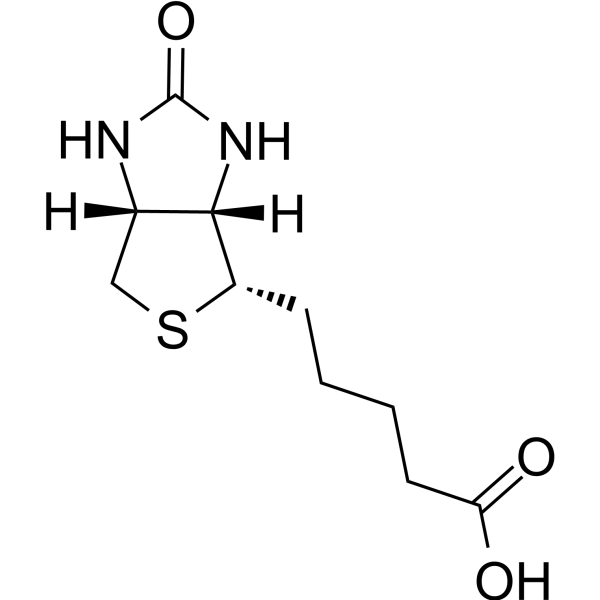
PDF) Effects of Biotin on survival, ensheathment, and ATP production by oligodendrocyte lineage cells in vitro
Pharmacokinetics and pharmacodynamics of MD1003 (high-dose biotin) in the treatment of progressive multiple sclerosis: Expert Opinion on Drug Metabolism & Toxicology: Vol 12, No 3

Could High-Dose Biotin Be Used as a Treatment for Multiple Sclerosis? - Multiple Sclerosis News Today

Andy Biotech on Twitter: "MedDay high dose biotin MD1003 Ph3 data in Progressive MS at #AAN15 http://t.co/FascW4Vfme @lomu_j @hydrogenblimp @Sport234a @jq1234t" / Twitter
Phase III Study of Experimental MS Therapy Meets Primary Endpoint, Results to be Presented at ANN Meeting

MedDay Pharmaceuticals Announces Full Patient Enrollment for MD1003 Phase III Clinical Trial (SPI2) in Progressive Multiple Sclerosis | Business Wire

High-dose pharmaceutical grade biotin (MD1003) in amyotrophic lateral sclerosis: A pilot study - EClinicalMedicine

PDF) MD1003 (High-Dose Pharmaceutical-Grade Biotin) for the Treatment of Chronic Visual Loss Related to Optic Neuritis in Multiple Sclerosis: A Randomized, Double-Blind, Placebo-Controlled Study