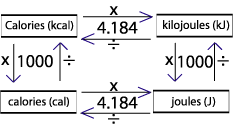
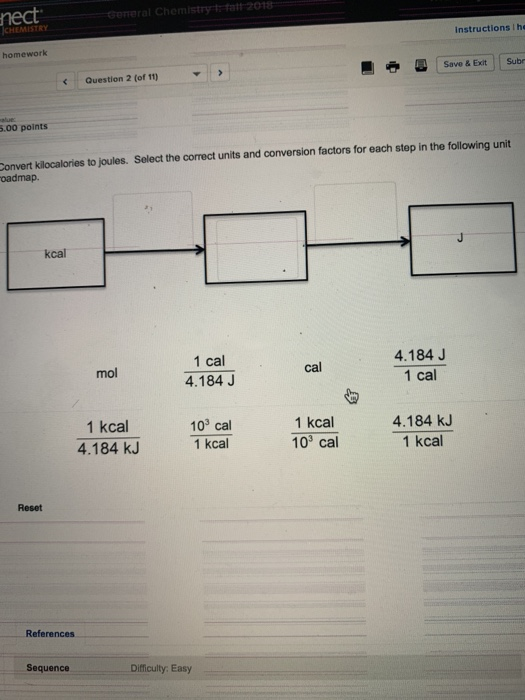
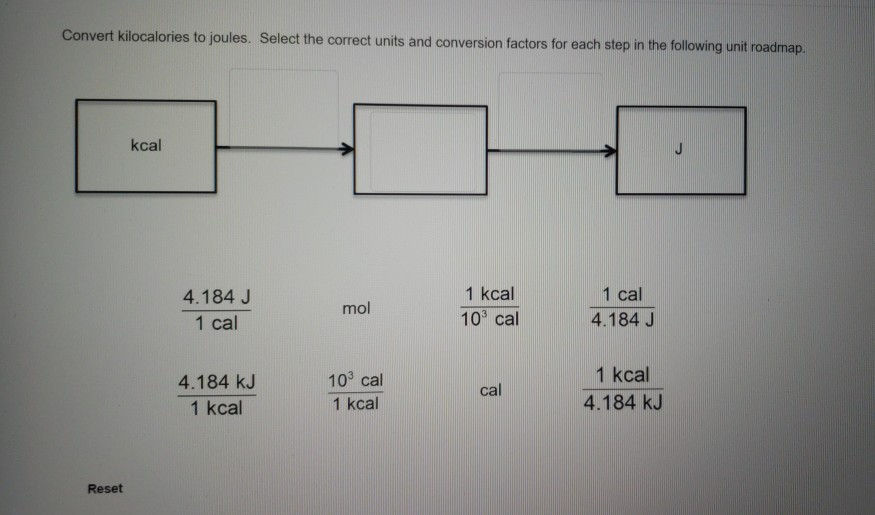
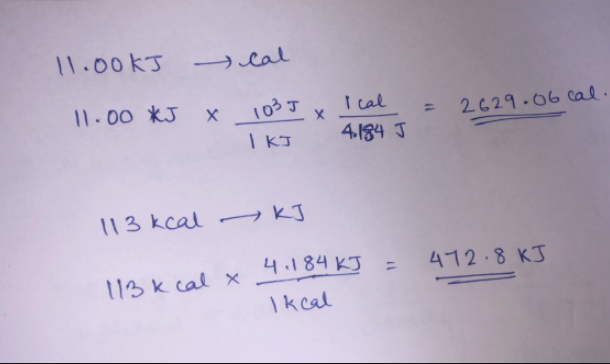Chapter 17 pgs q= m x c x T This is the equation for all Thermochemistry problems The Q is heat values in either calories or joules ( ppt download

The heat Q that must be supplied or removed to change the temperature of a substance of mass m by an amount ∆T is: Q = cm∆T where c is the specific. -