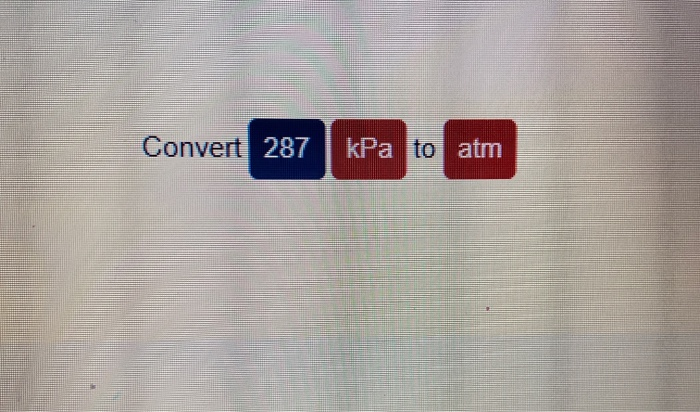
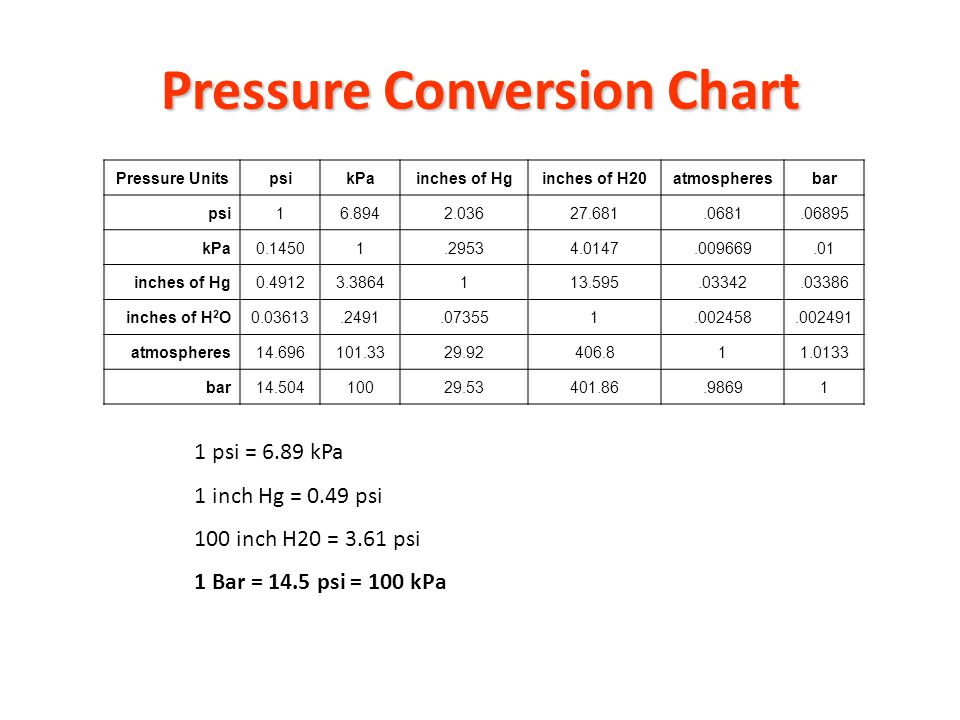
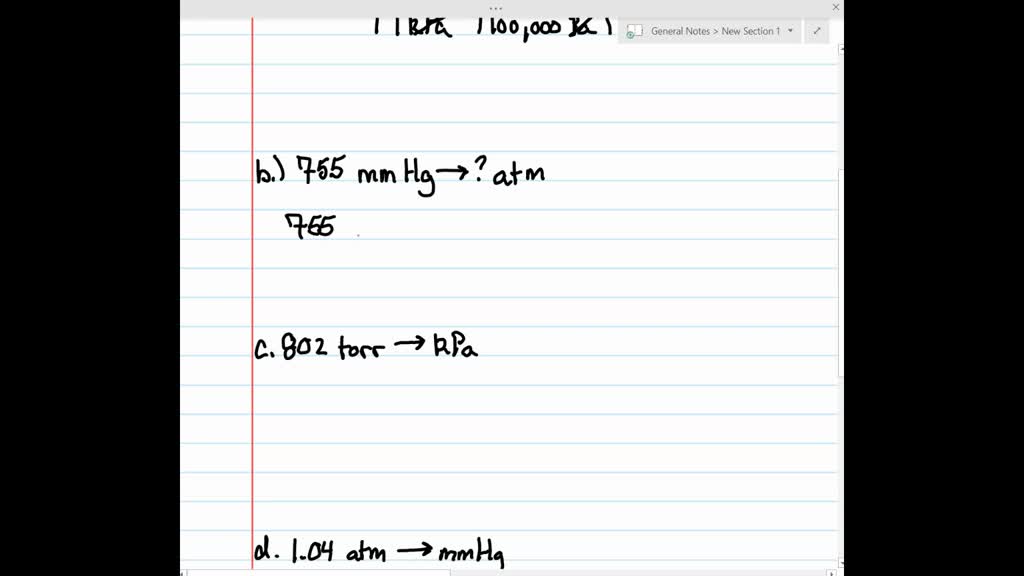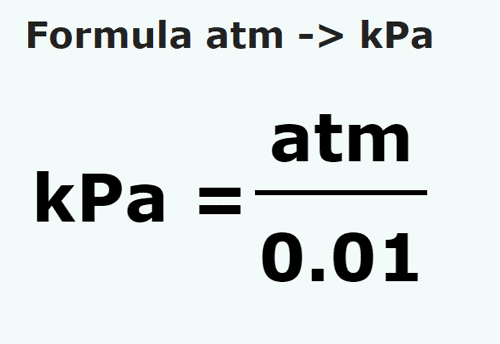✓ Solved: Gas pressures can be expressed in units of mm Hg, atm, torr, and kPa. Convert these pressure...

Journal 1)Convert the following pressures to pressures in standard atmospheres: A kPa B. 456 torr Conversions 1 atm=101.3 kPa= 760 mm Hg= ppt download

Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download