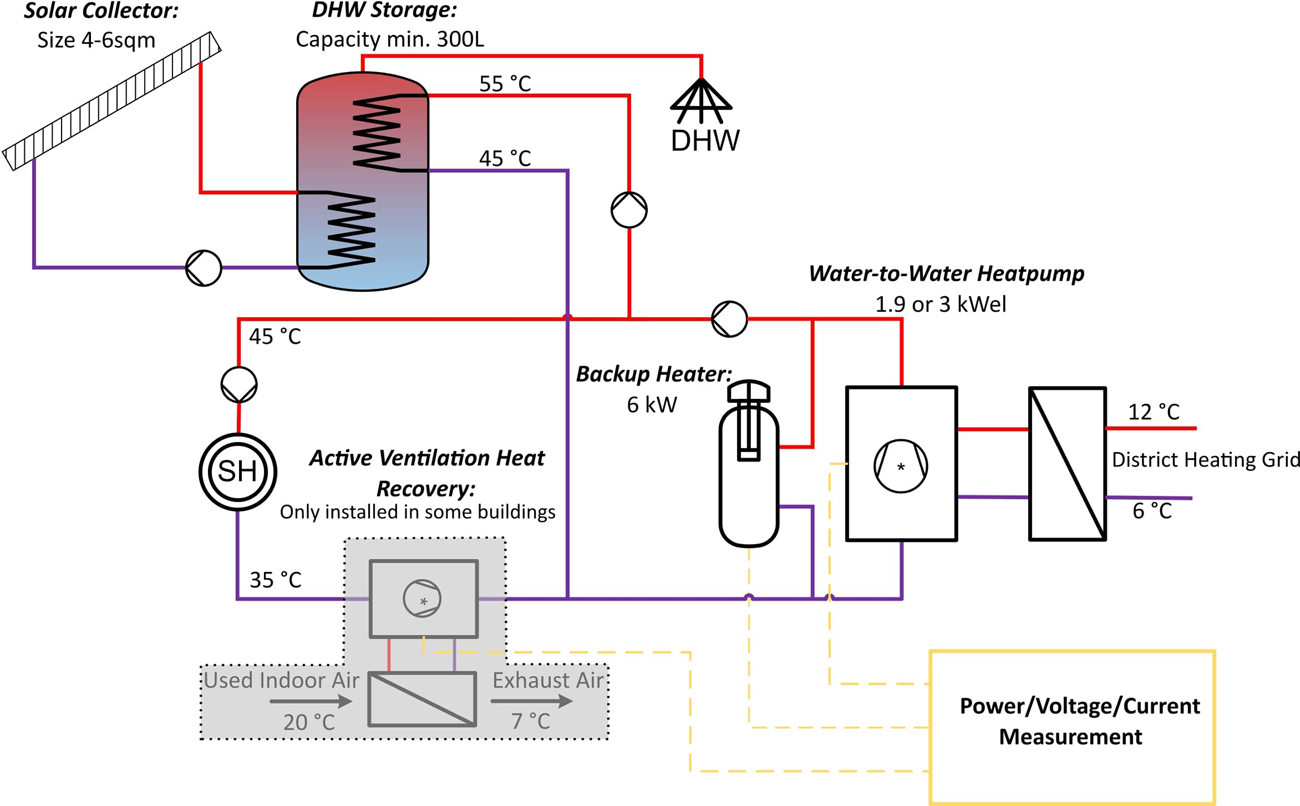
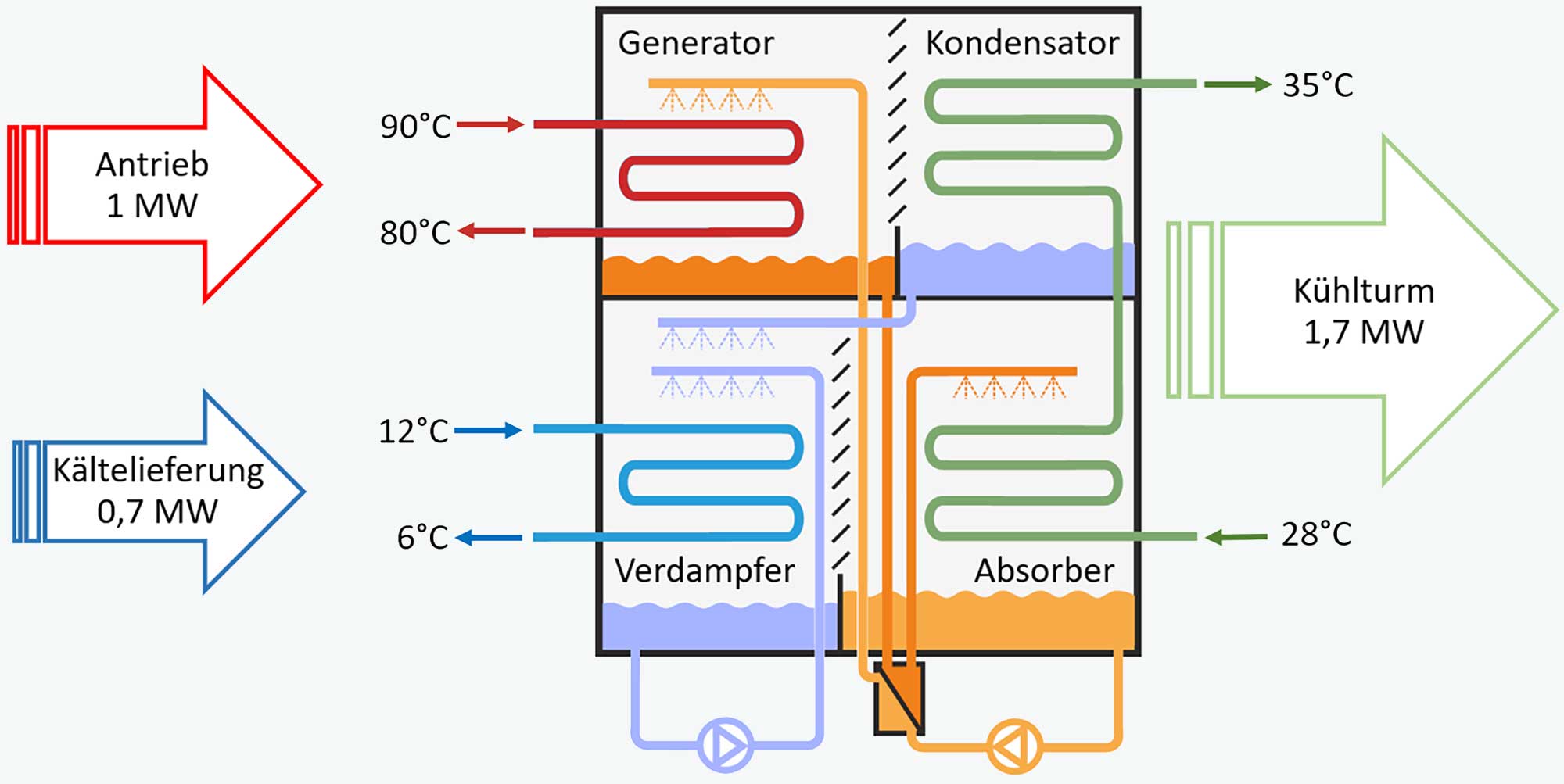
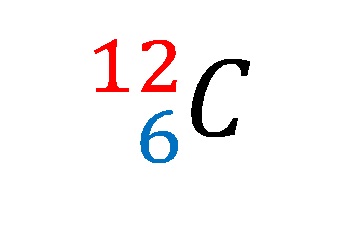Übungen - Ausmultiplizieren von Termen 2018 Aufgabe 1: Schreibe ohne Klammern. a) 3 · (4�� − 5) b) 8 · (5�� + 6�

Isótopos del carbono: 12 C (6 protones y 6 neutrones), 13 C (6 protones... | Download Scientific Diagram
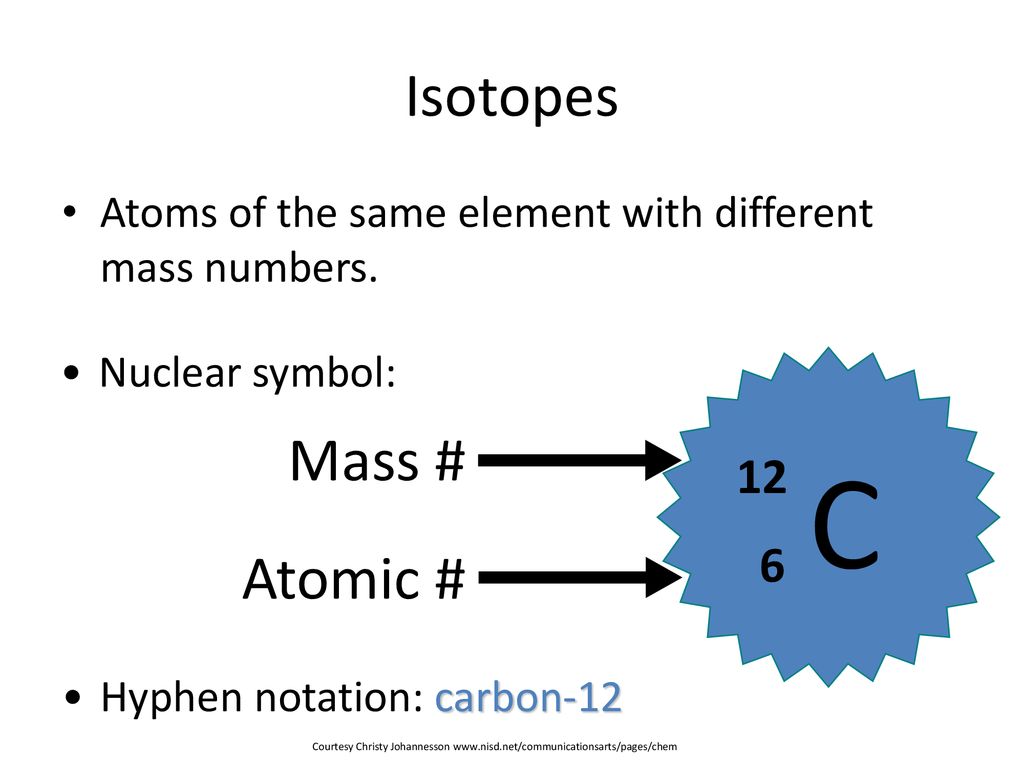
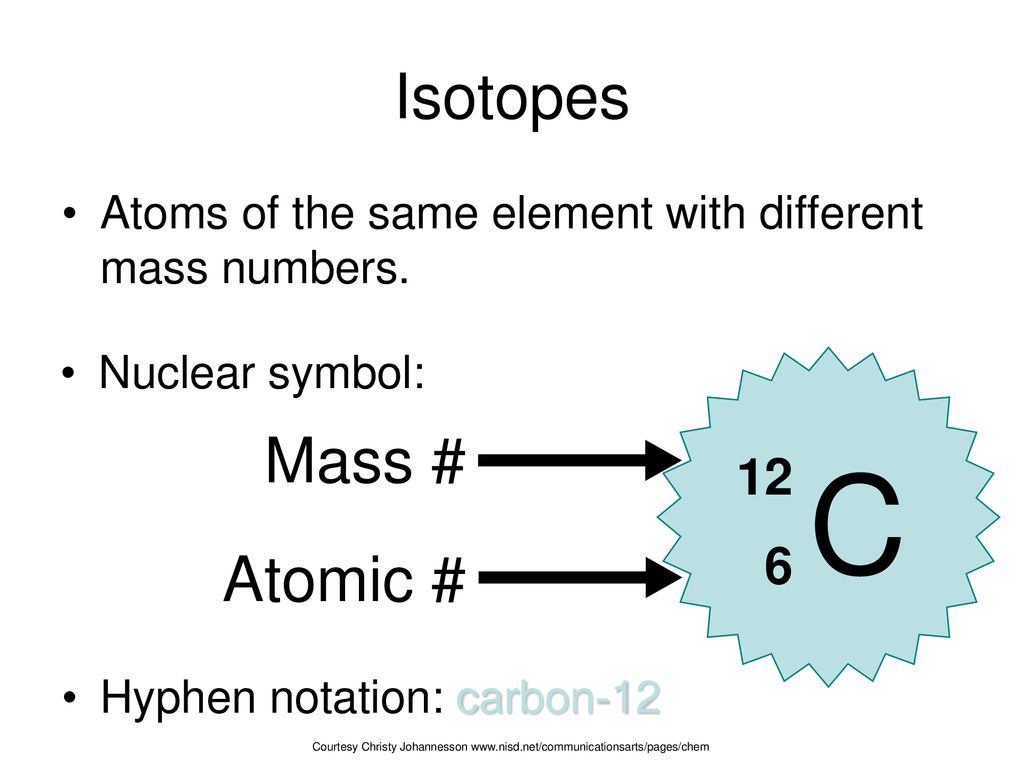
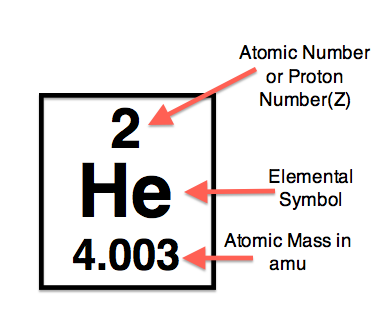
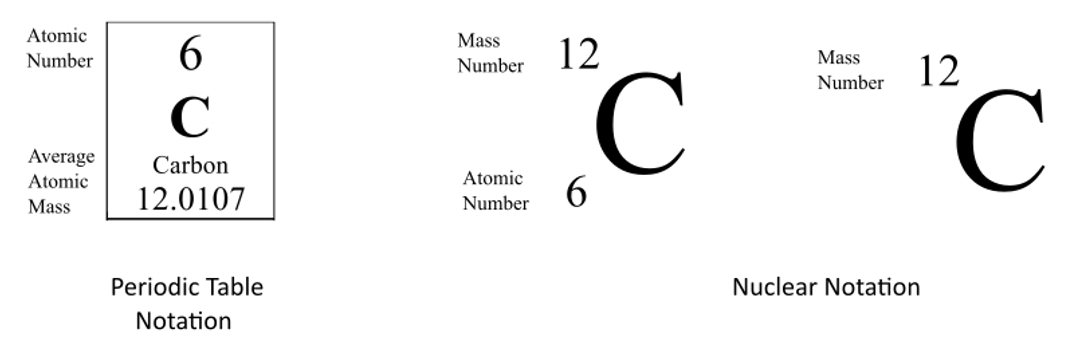
Symbols Contain the symbol of the element, the mass number and the atomic number X Mass number Atomic # protons + # neutrons mass number # protons. - ppt download
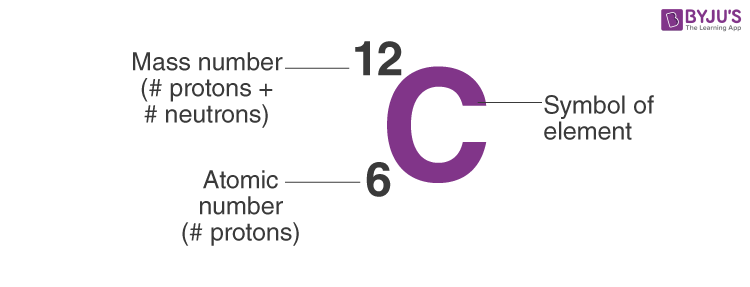
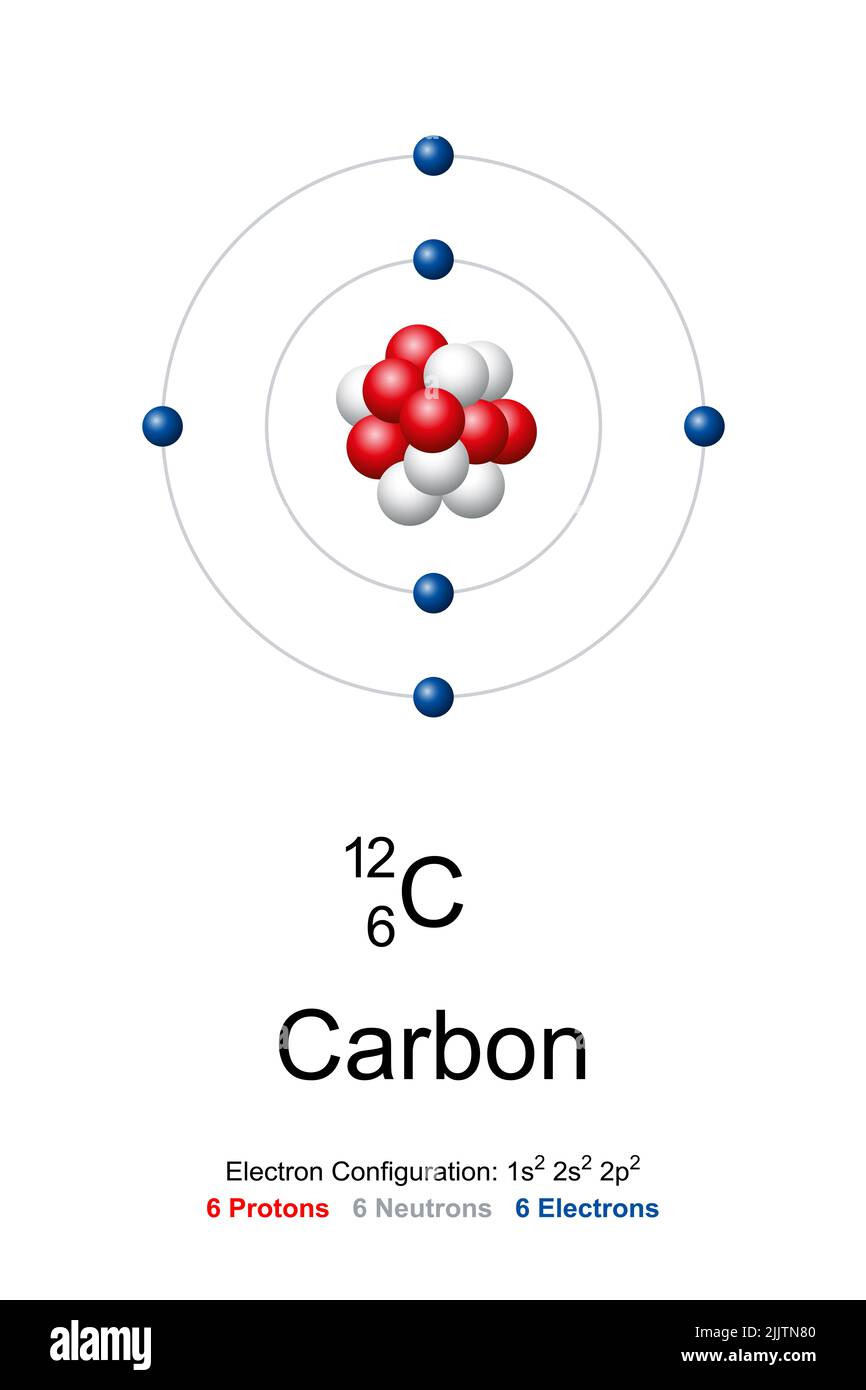
Carbon Protons - What are Protons, Number of protons in Carbon atom, and Uses of Carbon atom along with some FAQs

Atomic notation X A Z X = Symbol (C, Au) A = Atomic Mass Number = #nucleons (Protons + Neutrons) Z = Atomic Number = #protons C 12 6 Carbon A = 12, Z = - ppt download